By Liston Barber, Product Manager of Sterile Processing
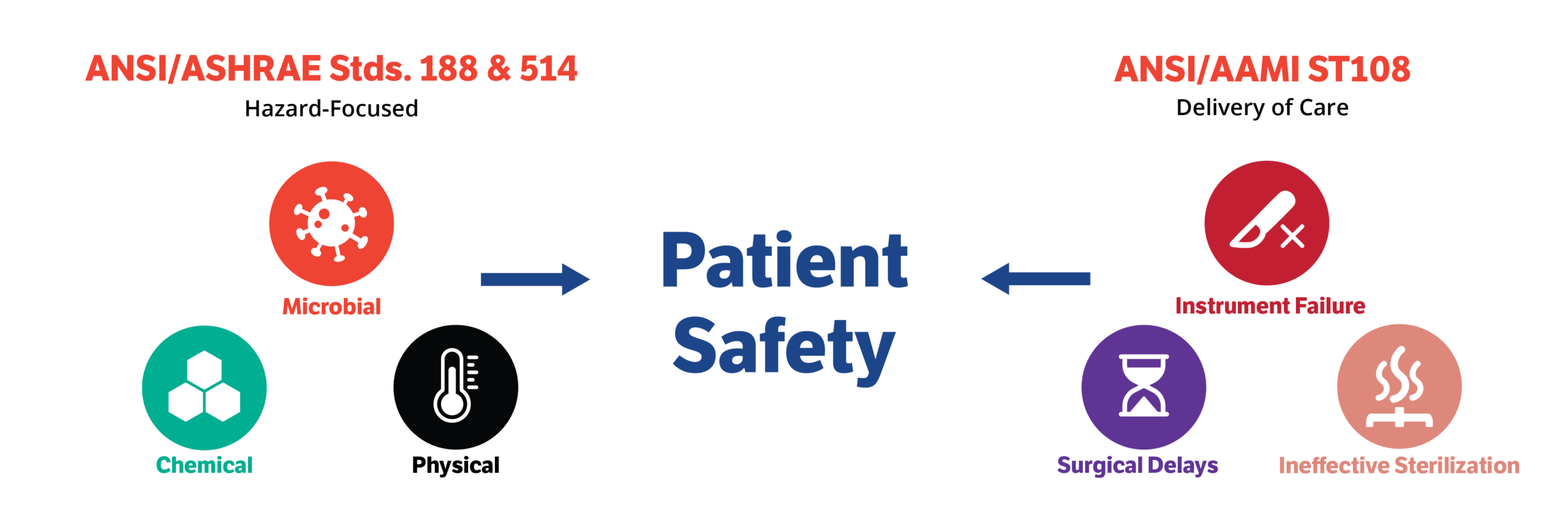
A frequent concern for Water Management Teams regarding ANSI/AAMI ST108 is what the standard says about risk — specifically, how to define it and how to reduce it. While most healthcare professionals are aware that water can pose significant risks in hospitals, many view this risk through the lens of ANSI/ASHRAE Standard 188, which focuses on Legionella, and ANSI/ASHRAE Standard 514, which expands the scope to include other waterborne pathogens, such as Pseudomonas. Compared to the aforementioned standards, ANSI/AAMI ST108 is prescriptive in terms of what to test for and when, but light on details for how to respond to elevated results, and which results matter.
First, let’s highlight the differences between how these standards define “risk” itself. ANSI/ASHRAE 188 is focused on the risk of patients acquiring a dangerous pathogen from building water. ANSI/AAMI ST108 looks at this same risk to patients, but also covers “process risk,” or the risk that your water quality may negatively impact the process of your SPD operations. This includes the risk of your instruments not being sufficiently cleaned, becoming corroded or stained, or your equipment failing. Your definition of risk should be expanded to include all of these considerations. If it can impact your ability to effectively reprocess instruments, it is a risk.
With the sheer volume and frequency of testing in alignment with ST108, it can be difficult to identify which test results to prioritize. Generally, elevated bacterial and endotoxin results are considered significant for “patient risk” because these are measures that may directly cause disease. Although sterilizers should be able to kill any bacteria on the instrument, they cannot effectively remove the endotoxin these bacteria leave behind, possibly leading to pyrogenic reactions in patients. Likewise poor critical water, steam feedwater, or condensate quality can lead to short equipment life or ineffective reprocessing, which represents a significant “process risk”. On the other hand, utility water chemistry may not be a major risk if it meets your equipment IFUs and the critical water is within standards, because the critical water final rinse should clean any minerals off the instrument that may have come from utility water. Remember that both types of risk impact patient safety.
The best way to manage these risks is to follow the ANSI/AAMI ST108 recommendation of building a Water Management Program for sterile processing. The standard suggests involving sterile processing stakeholders in your multi-disciplinary Water Management Team (WMT) and monitoring water quality. I discussed this on our latest webinar, “From the Field: Lessons Learned from Implementing ANSI/AAMI ST108,” with Robin Quinn, one of our Business Development Managers at Phigenics. Robin and I emphasized some of what we consider best practices for a WMT, such as quarterly meetings, annual assessments, and using trending data to ensure a system is effective.
If you have questions about ST108 or Water Management for Sterile Processing in general at your facility, please feel free to contact me at lbarber@phigenics.com.
